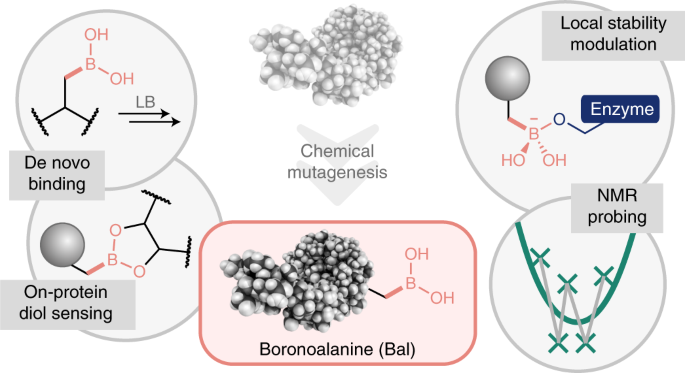
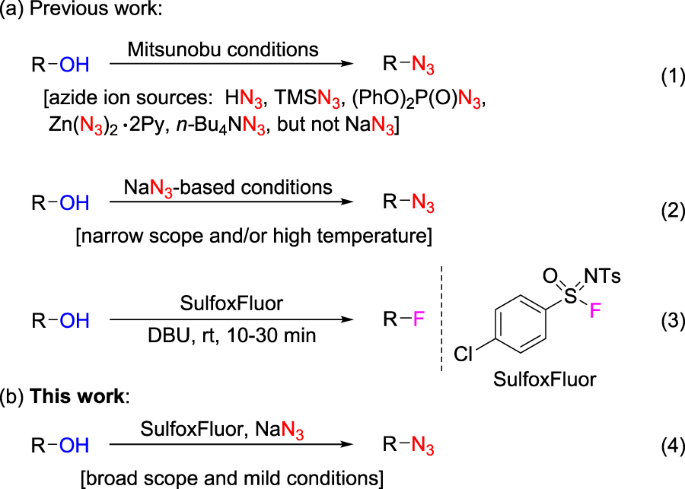
H3BO3 Lewis Structure: How to Draw the Lewis Structure for B(OH)3
$ 43.50
-
By A Mystery Man Writer
-
-
4.6(248)

Product Description
A step-by-step explanation of how to draw the B(OH)3 Lewis Dot Structure (Boric Acid, also called H3BO3).Note that Boron (B) is an exception to the octet rul

SOLVED: Boric acid, H3BO3, also written as B(OH)3, acts as a weak acid in water. However, it does not do so by loss of a hydrogen ion. Instead, it acts as a

NCERT Exemplar Class 11 Chemistry Solutions Chapter 11 - The P Block Elements

Basicity and structure of H3BO3 Boric Acid, Inorganic Chemistry, NEET

Boric acid is weak monobasic acid and acts as Lewis acid

Lewis Dot Structure of H3BO3 or B(OH)3 (Boric Acid)

H3BO3 (Boric Acid) Lewis Structure

Basicity and structure of H3BO3 Boric Acid, Inorganic Chemistry, NEET
How to determine the Lewis dot structure of H3BO3 - Quora

Basicity and structure of H3BO3 Boric Acid, Inorganic Chemistry, NEET